 Activity series of some of the more common metals, listed in. 2: Elements categorized into metals, non-metals and metalloids. The oxidation number of an element in this group can range from +5 to -2, depending on the group in which it is located. 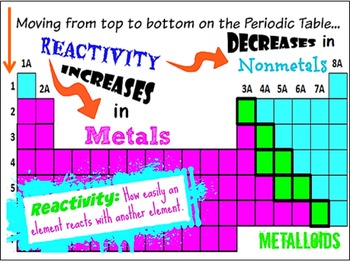
It is used to determine the products of single displacement reactions, whereby metal A will replace another metal B in a solution if A is higher in the series. The physical properties of metalloids tend to be metallic, but their chemical properties tend to be non-metallic. So, the more the number of inner electrons, the more will be a shielding effect and more easily the outermost electron can escape from the atom. The reactivity series is a series of metals, in order of reactivity from highest to lowest. The shielding effect is the effect produced by inner orbital electrons that come and hinder the path of nuclear charge and thus shields the outermost electrons. Hence, the reactivity of alkali metals increases on moving down the group. Thus these valence electrons are now easily available to take part in a reaction and the reactivity of the element is said to be increased. The activity series is a list of elements in decreasing order of their reactivity. The outermost electrons become farther away from the nucleus.Īs a result, the shielding effect increases, and the valence electrons are more loosely bound to the nucleus. Four major factors affect reactivity of metals: nuclear charge, atomic radius, shielding effect and sublevel arrangement (of electrons). As we move down the group, the atomic number and number of shells also increase. The elements within the same group of the periodic table tend to exhibit similar physical and chemical properties. The alkali metals have the largest atomic radii and the lowest first ionization energy in their respective periods.Īlthough all alkali metals have one valence electron in their valence shell, there are slight differences in the reactivity order of elements on going down the group. The properties of the alkali metals are similar to each other as expected for elements in the same family. All alkali metals have their outermost electron in the s-orbital and thus they correspond to the s-block. These elements constitute the first group of the periodic table. The alkali metals are lithium (Li), sodium (Na), potassium (K), rubidium (Rb), cesium (Cs), and francium (Fr). The largest atomic radius of elements in their period. 
Alkali metals tend to lose this electron when they participate in reactions. Highly reactive, with reactivity increasing moving down the group. All alkali metals have only one electron in their valence shell. A great Starter for ten activity to engage your students with trends in the periodic table by focusing on group 2, group 3 and group 7. a microscale practical into the extraction of copper, where students can engage with the experiment and build on their practical chemistry skills. 
As chlorine can easily accept an electron as compared to phosphorus and sulphur, the chemical reactivity increases from phosphorus to chlorine.Hint: In chemistry, reactivity is a measure of how readily a substance undergoes a chemical reaction. The extraction of copper: a microscale version. Chlorine (Cl) has 7 valence electrons and needs only one more electron to complete its octet. But, at the far right are the noble gasses, and they are almost completely. Each element going from left to right tends to be more reactive. Reactivity: The reactivity of the elements increases going from left to right on the periodic table. Sulphur (S) has 6 valence electrons and needs 2 more electrons to complete its octet. The periodic table can be used to determine the following properties of materials: Atomic number (Z). The most vigorously reactive metals of the periodic table are alkali metals. Phosphorus (P) has 5 valence electrons and needs 3 electrons to complete its octet. In chemistry, reactivity is the impulse for which a chemical substance undergoes a chemical reaction, either by itself or with other materials, with an overall release of energy. Moving further right in the period towards non-metals, the chemical reactivity again gradually increases. This is because the number of valence electrons increases, making it difficult to lose electrons. The chemical reactivity gradually decreases as we go to aluminium (Al) and silicon (Si). On moving from left to right in a period of the periodic table, the chemical reactivity of the elements first decreases and then increases.įor example, in the third period elements comprising of Na, Mg, Al, Si, P, S and Cl, sodium (Na) is very reactive as it has one valence electron, and can therefore, lose its electron easily. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
